Here's the problem with electricity: it costs 5 cents to make a kilowatt-hour, but it costs $150 worth of batteries to store a kilowatt-hour. And it's only that cheap if you use lead-acid batteries, which are (a) very heavy; and (b) made with toxic materials.
And that's the rub for electric cars, because the heavier the car is, the more batteries it needs to move. So if you use cheap lead-acid batteries for electric cars, the car weighs two tons, and needs more power, which makes it heavier, so it needs more power, which makes it heavier.
So instead you end up using lighter lithium batteries so the car has reasonable range and performance -- and the battery pack alone costs $24,000 (instead of the $8000 that lead-acid would cost with worse performance). Yuck.
Wouldn't it be nice if there were a battery that was as light as lithium, and as cheap as lead-acid? Wouldn't it be nice if it were made of completely non-toxic materials -- so safe you could eat them? Wouldn't it be nice if it were being manufactured right now, right here in the USA?
Wouldn't it be odd if all that were true, and you've never even heard of it?
Well maybe there's a good reason for that. The battery in question -- the aqueous sodium battery -- is actually too good for automotive applications! It's so good, and has such an incredible lifespan, that the manufacturer is looking only to hard-core industrial uses for its first customers, like load-leveling for renewable energy systems. Not that that's a bad thing. But I'm ahead of my story.
Our hero is Jay Whitacre, an engineering prof at Carnegie-Mellon U. in Pittsburgh. Back in 2007, a colleague gave him a challenge. You've been working on lithium-ion batteries, said the colleague, but what the world really needs is a battery that's dirt cheap. Find that, and you can change the world.
Whitacre took up the challenge. A battery is composed of three components: two electrodes, separated by an electrolyte. The electrolyte won't allow electrons (that's electricity) to pass through, but it will allow ions -- electrically charged atoms or molecules -- to pass through. The cheapest possible electrolyte is water, or something water-based, so that's where Whitacre started. But water alone isn't an electrolyte, because the electrolyte also needs ions -- in fact, it needs positively-charged ions called cations. In a lead-acid battery, those ions are hydrogen ions, which is why the water in the lead-acid battery is acid: when you put hydrogen ions in water, you get acid.
But Whitacre had another idea. Instead of hydrogen cations, he went with sodium cations. Sodium ions are found in plain saltwater, and unlike metallic sodium, the ions are completely harmless. Most importantly, they're harmless and non-corrosive to metal, which make up the electrodes.
For the cathode (the positive electrode), Whitacre tried several metals, and finally settled on magnesium manganese oxide, because it's the cheapest metal oxide you can find. He found a particular arrangement of atoms on the surface that worked particularly well. For the other electrode, the anode, he used plain carbon -- even deriving the carbon from molasses or syrup.
After a few years and a couple of patents, Whitacre formed a startup, Aquion Energy, to manufacture the aqueous sodium battery. Aquion is currently building first-run batteries for beta-test customers, and is looking for larger manufacturing space to expand during 2012. They're moving fast, and have already attracted significant venture capital and a grant from the DOE in 2009.
* * * *
So why is the aqueous sodium battery "too good" for vehicles? Let's start with this chart, adapted from Shukla, Arico, & Antonucci 2001, Renewable and Sustainable Energy Reviews 5, 137–155. I've superimposed the data from the aqueous sodium battery in red. ("Na" is the chemical symbol for sodium, for those who wondered.)
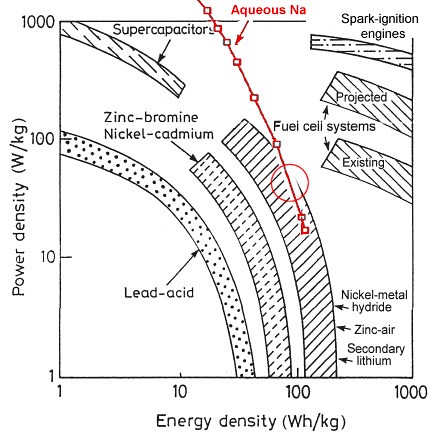
Basically, technologies farther to the right on the chart are better for cars: more energy for a given weight. Over on the left, lead-acid batteries make a poor showing. Ni-Cad is a definite improvement; and farther right is a grouping of lithium, nickel-metal-hydride, and zinc-air, and that's about where aqueous sodium falls too. The red circle is about where on the aqueous sodium curve you would expect to run a commuting electric car: about two hours total discharge time, which corresponds to about 90 Watt-hours per kilogram. Lithium does a bit better, but not by much.
Actually, if you look only at a diagram like this, the best technology would seem to be zinc-air. It's way over to the right of that diagonally-hatched area, at about 140 to 150 watt-hours per kg, and zinc-air batteries are about as cheap as lead acid to buy. So why aren't EV makers rushing to use zinc-air batteries?
The answer is what's not on this chart: zinc-air batteries have very short lifespans. You can only go though about 200 charge/discharge cycles before they're done. For a commuting EV charging and discharging five days a week, that means you'd have maybe a year before you'd have to replace your battery pack. And yeah, zinc-air is cheaper than lithium, but who wants to replace an $8000 battery pack every year?
And that's where aqueous sodium batteries really shine. Because while zinc-air batteries can go 200 cycles, and lead-acid can go 250 to 500 cycles, and lithium can go 500 to 1000 cycles before replacement, aqueous sodium batteries have very long lifespans. So long that not even Aquion knows for sure how long they will last. The numbers they're guessing are truly staggering: about 10,000 cycles. Those sodium ions are just too mild to damage those electrodes no matter how often they shuffle back and forth.
And that's also why aqueous sodium batteries are too good for cars. Let's say you build a typical electric commutermobile, with a hundred-mile range. And let's say your commute is 100 miles, so you fully charge and discharge your EV on every commute. How long would an aqueous sodium battery pack last in your vehicle?
One MILLION miles.
That's assuming you get 10,000 cycles out of your battery pack. But even if you don't, it's clear that the battery pack would last several times longer than the vehicle itself. And that's the problem: the battery pack is the most expensive part of the EV. That's where most of the capital costs go. And when the vehicle has reached the end of it's life, the battery pack will still be a teenager, with a looooong life ahead. So if you were to buy a new electric car with an aqueous sodium battery, you would actually be spending a lot more on capital costs than you really should, because that expensive battery just ain't gonna wear out in time with the rest of the car.
The economics of the market in this situation are fairly complex. When the car is a junker, can you remove the battery pack and sell it used? Presumably. But wouldn't the buyer want to know how many cycles the pack had on it, just like the buyer of a used car wants to know the mileage? How would the buyer know if the battery pack had already powered through the lifetimes of two cars, or four, or six? And how could you actually track that number that in a non-corruptible way? Beats me.
But I guess that's what the comments are for.
And I guess that's why Aquion is concentrating on heavy-duty, non-mobile industrial applications. If you can make it there, you can make it anywhere -- except, maybe, in cars?
h/t Smart Planet for this video interview with Jay Whitacre.


