As we discover thousands of exoplanets, the interest in life outside our blue marble has renewed. The various searches for earth-like planets focus on planets with liquid water and assume that life is based on carbon. Is this mere prejudice, a failure to think outside the box? Why do we think life will be carbon based, and how much wiggle room is left for other lifeforms?
We begin with the assumption that chemistry is universal. If in the unlikely event we travel to another galaxy, we expect to find the same palette of atoms that we enjoy here: hydrogen, oxygen, carbon and so on. Chemistry arises out of the properties of atoms, and so we expect the possible range of chemistries also to be the same around any star and in any galaxy.
Given that, is there anything unusual about carbon that favors life? Carbon can make four stable bonds. This is unusual but not unique, as silicon, tin and lead can also do this. Making four covalent bonds allows for a variety of forms not possible with fewer: long chains, rings, branched compounds, the whole zoo that plagues first semester organic chemistry students.
But carbon can do more than make four bonds: it can make stable double bonds, it can take on different oxidation states in the same molecule, and it can form stable bonds to a number of other atoms. Thus we have the splendor of a DNA molecule, with regions of sugar phosphate esters in chains and squiggly bits on the inside of aromatic rings that stack and expose hydrogen bonding sites, like badly made zippers.
This sounds merely theoretical to many; surely it's just a lack of imagination that we can't think of other compounds? Is there any way to test the spontaneous variety of compounds?
As it happens, there is. In the last few decades spectroscopy has grown and developed to the point we can explore the composition of interstellar space and dust clouds. Such studies allow a tally of what compounds have been found in space, formed simply by the inherent tendency to structure.
This table of molecules in space is actively maintained and has, if not an exhaustive, a representative sample of all compounds that have been detected in space. We can use it to gain a sense of how large a molecule can be formed from which atoms. Size is a necessary property for complexity, if we are thinking about molecules that can support life. How many molecules of each size have been detected, how many of those are organic, and what percentage of that size is organic? 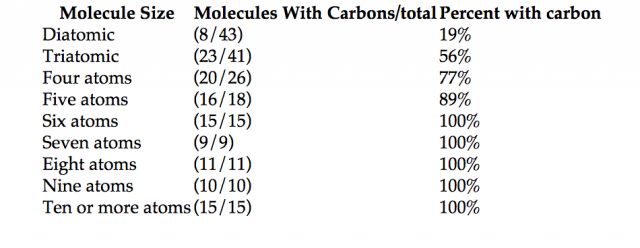
All the molecules with six or more atoms are organic molecules. This is a strong measure of the inherent tendency to form structures, based on the chemistry of carbon and the abundance of the atoms. It seems clear that the strategy to look for carbon-based life is the way to go.
Oh, and the raspberries? Here you go.
Silicon-based life at TV Tropes
The Chemistry of Space - NASA astrobiology portal
Meteorite with organic molecules - Murchison meteorite
In this study, 14,000 specific compounds, including 70 amino acids, were identified. But this number appears to be the tip of the iceberg; the meteorite probably contains millions of different organic compounds. More detailed analysis will now be carried out.
More about the
Murchison meteorite
Complex organic matter from novae
Simple organics on Mars and on comet Philae


